AACR 2026 – Qilu challenges Torl in Claudin6
The company’s conjugate QLS5132 has produced intriguing, but early results.
The company’s conjugate QLS5132 has produced intriguing, but early results.

Several Claudin6 projects have fallen short of expectations, but Qilu is the latest to take aim at this target with its ADC contender QLS5132. At an AACR plenary session on Sunday the company reported a best response rate of 50% in platinum-resistant ovarian cancer, though a closer look shows a confirmed response rate of just 28%.
Still, Qilu could give Torl Biotherapeutics’ ixotatug vedotin a run for its money, particularly as the latter has largely shown promise in Claudin6-positive disease. One of the most intriguing aspects of Qilu's presentation are reports of responses among patients with undetectable Claudin6 expression levels.
There are reasons to be cautious about the first-in-human results for QLS5132, notably that they come from a China-only trial, and involve just 18 evaluable patients. In addition, the number encompasses various doses, including 6.4mg/kg, which saw a dose-limiting toxicity.
However, although 6.4mg/kg was the highest dose tested, it didn’t appear to be the most efficacious. That honour goes to 4.8mg/kg, which produced five partial responses (three confirmed) among nine patients, giving a 33% ORR, or 56% best response rate.
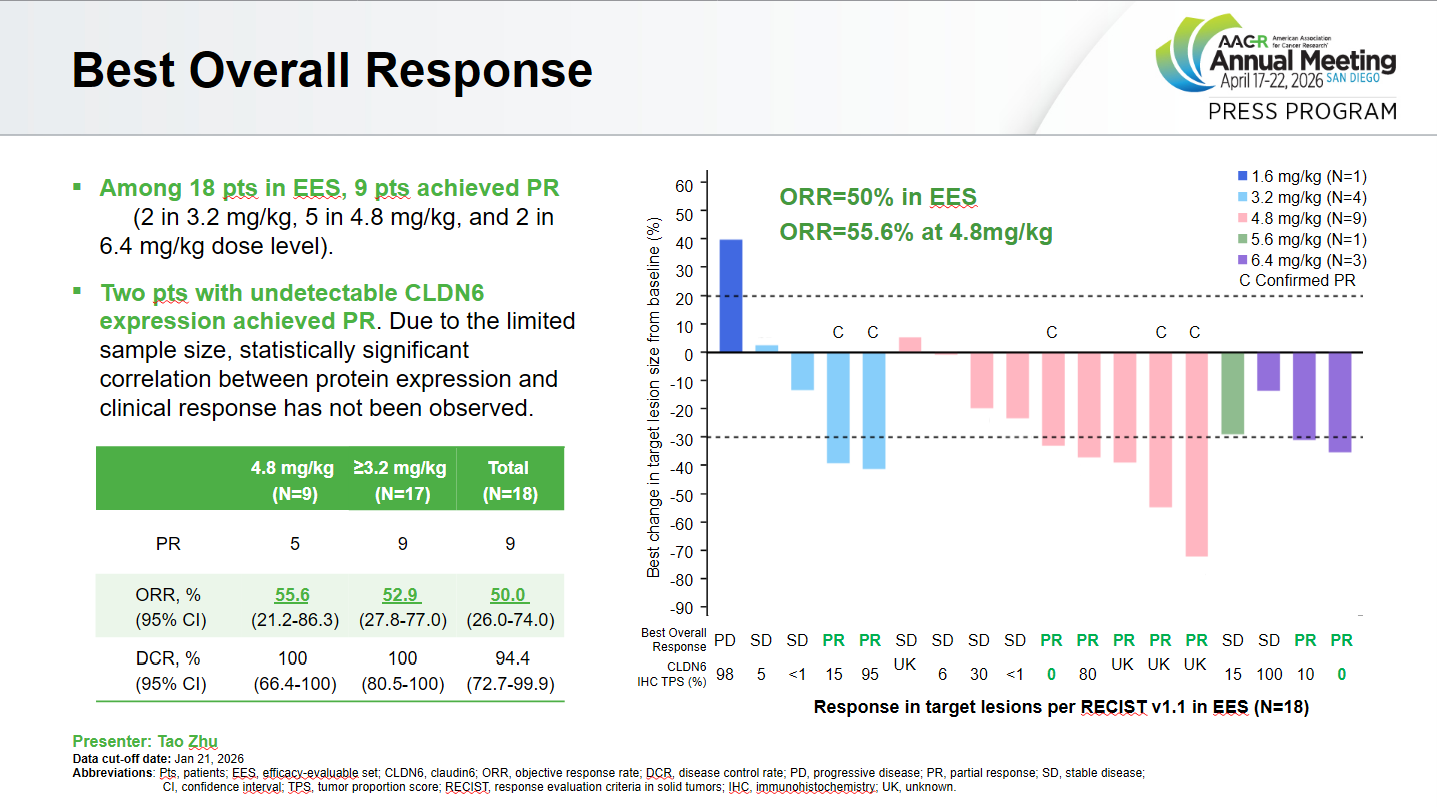
Notably, two patients with undetectable Claudin6 levels achieved partial responses. Overall, only 43% of the 28 enrolled PROC patients were Claudin6-positive by immunohistochemistry testing.
Meanwhile, at last year’s ESMO meeting Torl’s ixotatug vedotin showed a confirmed ORR of 50% across 26 platinum resistant ovarian cancer patients in its phase 1 study, Catalina-1 – but these were all Claudin6-positive, and this number only came from three doses: 2.4mg/kg, 3.0mg/kg and 3.4mg/kg.
Ixotatug vedotin's phase 2 Catalina-2 trial is focused on Claudin6-positive PROC.
Cross-trial comparison of Claudin-6-targeted ADCs in PROC
| QLS5132 | Ixotatug vedotin (TORL-1-23) | |
|---|---|---|
| Company | Qilu Pharmaceutical | Torl Biotherapeutics |
| ADC details | Topo1 inhibitor payload, DAR 8 | MMAE payload, DAR 4 |
| Study | China ph1 | US ph1 Catalina-1 |
| Venue | AACR 2026 | ESMO 2025 |
| Cutoff date | 21 Jan 2026 | 7 Apr 2025 |
| Confirmed ORR | 28% (5/18) | 50% (13/26)* |
Note: *Claudin6+ve PROC only at 2.4-3.4mg/kg. Source: AACR & ESMO.
Ixotatug vedotin uses an auristatin payload, while QLS5132 employs a more fashionable topoisomerase 1 inhibitor. Professor Tao Zhu of Zhejiang Cancer Hospital, who presented the data at AACR, suggested that tumour heterogeneity and a “potent bystander effect” could result in activity with QLS5132 even in cells with low or no Claudin6 expression.
As for side effects, 32% of patients receiving QLS5132 experienced a grade 3 or higher treatment-related adverse event, with one serious event. However, no patients discontinued or died owing to treatment-related events, and there were no cases of interstitial lung disease, ocular toxicity or febrile neutropenia. The dose-limiting toxicity at 6.4mg/kg was a platelet count decrease; the potential go-forward doses are 4.8mg/kg and 5.6mg/kg.
Qilu’s phase 1 trial also includes cohorts in NSCLC, gastric and GEJ cancers, and other solid tumours, including those with low Claudin6 expression. The group is also starting a phase 1/2 combination trial in China.
Although Torl continues to advance ixotatug vedotin, other Claudin6-targeting projects have run into difficulties. Daiichi Sankyo abandoned its ADC, DS-9606, earlier this year, while BioNTech last year terminated one trial of its Car-T therapy BNT211, and reported disappointing results with its mRNA-encoded T-cell engager BNT142.
Others also developing Claudin6-targeting ADCs include Axcynsis Therapeutics with AT03-65, and PrimeLink BioTherapeutics with PLB-002, both in phase 1.
12