ASCO-GU 2026 – gauging Welireg’s double win
Welireg looks headed to regulators in second-line and adjuvant kidney cancer.
Welireg looks headed to regulators in second-line and adjuvant kidney cancer.

Merck & Co’s quest to expand Welireg’s reach beyond heavily pretreasted kidney cancer could at last come to fruition, with full data from the Litespark-011 and 022 trials confirming the drug’s benefit in the post-PD-(L)1 and adjuvant settings respectively.
The findings, revealed today at late-breaking presentations at ASCO’s Genitourinary Cancers symposium, come after both studies were toplined as positive last October, with full data now confirming their clinical meaningfulness. The results also reiterate the importance of the known toxicities of Welireg, a HIF-2α inhibitor whose current label warns of anaemia and hypoxia.
Importantly, however, the toxicities don’t appear to have overshadowed either trial. Memorial Sloan Kettering’s Dr Robert Motzer, the lead author of Litespark-011, went as far as to say that the toxicities weren’t worse with Welireg than control, but simply that they were different.
Litespark-011 concerned kidney cancer patients who had relapsed on anti-PD-(L)1 therapy, who currently can be given kinase inhibitors like Exelixis’s Cabometyx or Merck/Eisai’s Lenvima. The aim was to improve on this by combining Welireg with Lenvima, and in the control cohort patients got Cabometyx.
On the co-primary endpoint of PFS the Welireg combo scored a clear win, with a 4.1-month median increase and reduction in risk of progression or death of 30% (p=0.00007), Motzer said. When Merck toplined Litespark-011 it said the other co-primary of OS had only shown a numerical benefit, but Motzer revealed that this benefit amounted to a median of seven months.
Overall survival (co-primary endpoint) in Litespark-011
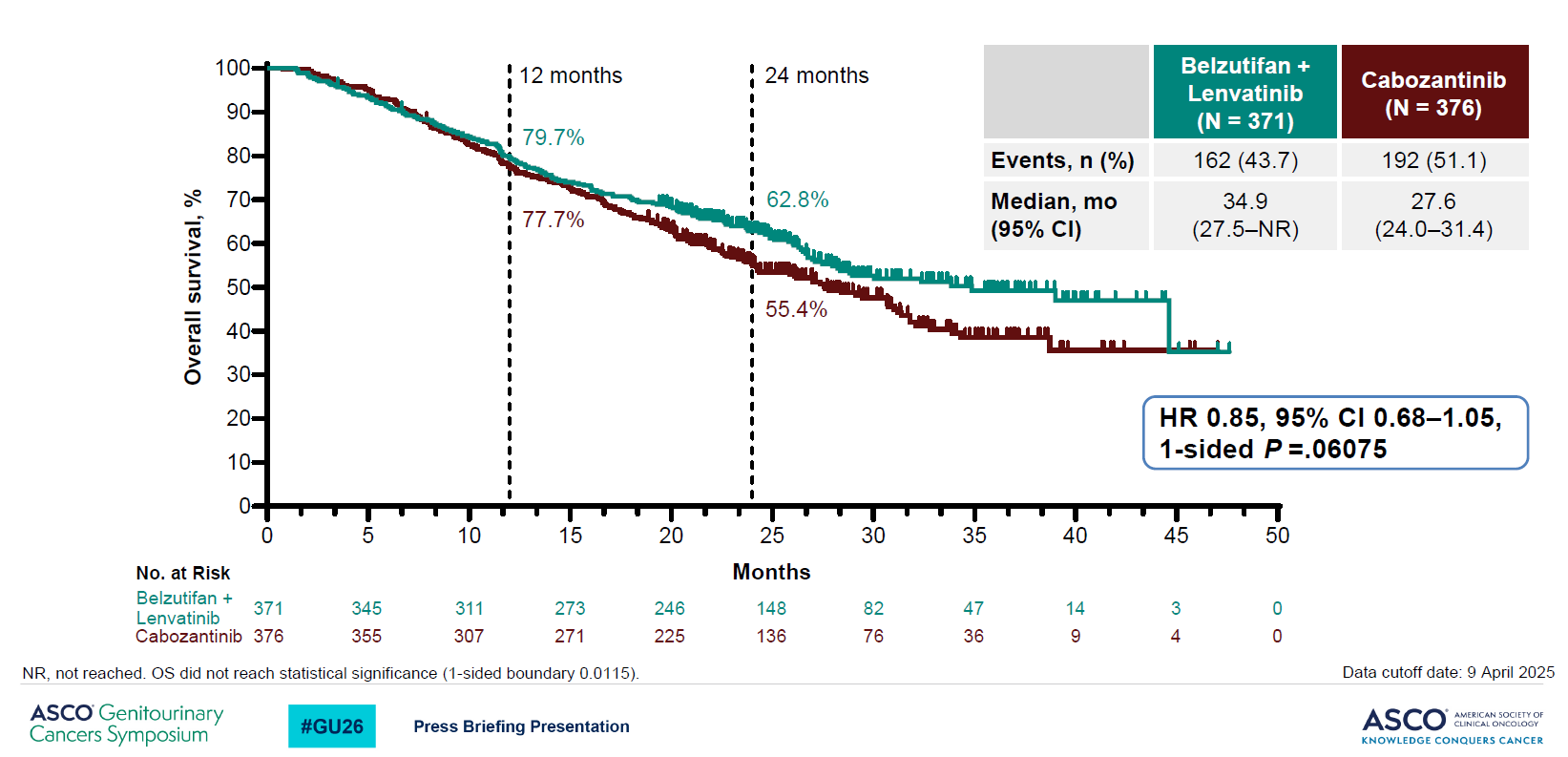
Moreover, the survival curves showed promising separation and a hazard ratio for OS of 0.85, albeit with a confidence interval upper bound over 1.00. At 0.06 the p value missed a tough boundary of 0.01, but Motzer said these data related to a second interim analysis, and a final OS read would come later.
Whether Litespark-011 has any chance to hit significance on OS remains an open question, but one positive sign is that when the FDA approved Welireg for renal cell carcinoma patients progressed on PD-(L)1 and VEGF inhibition it did so with the backing only of a PFS benefit.
As for adverse events, Motzer said there was a “trade-off”, with Welireg contributing anaemia and hypoxia, but Cabometyx resulting in more diarrhoea and skin toxicities. He rejected the suggestion that Welireg caused an increase in tox, but accepted that Welireg might not be the best choice in patients with co-morbidities including anaemia or hypoxia.
“For most fit patients I would prefer [the Welireg combo] over Cabometyx,” he told a pre-ASCO-GU press briefing.
Adjuvant use
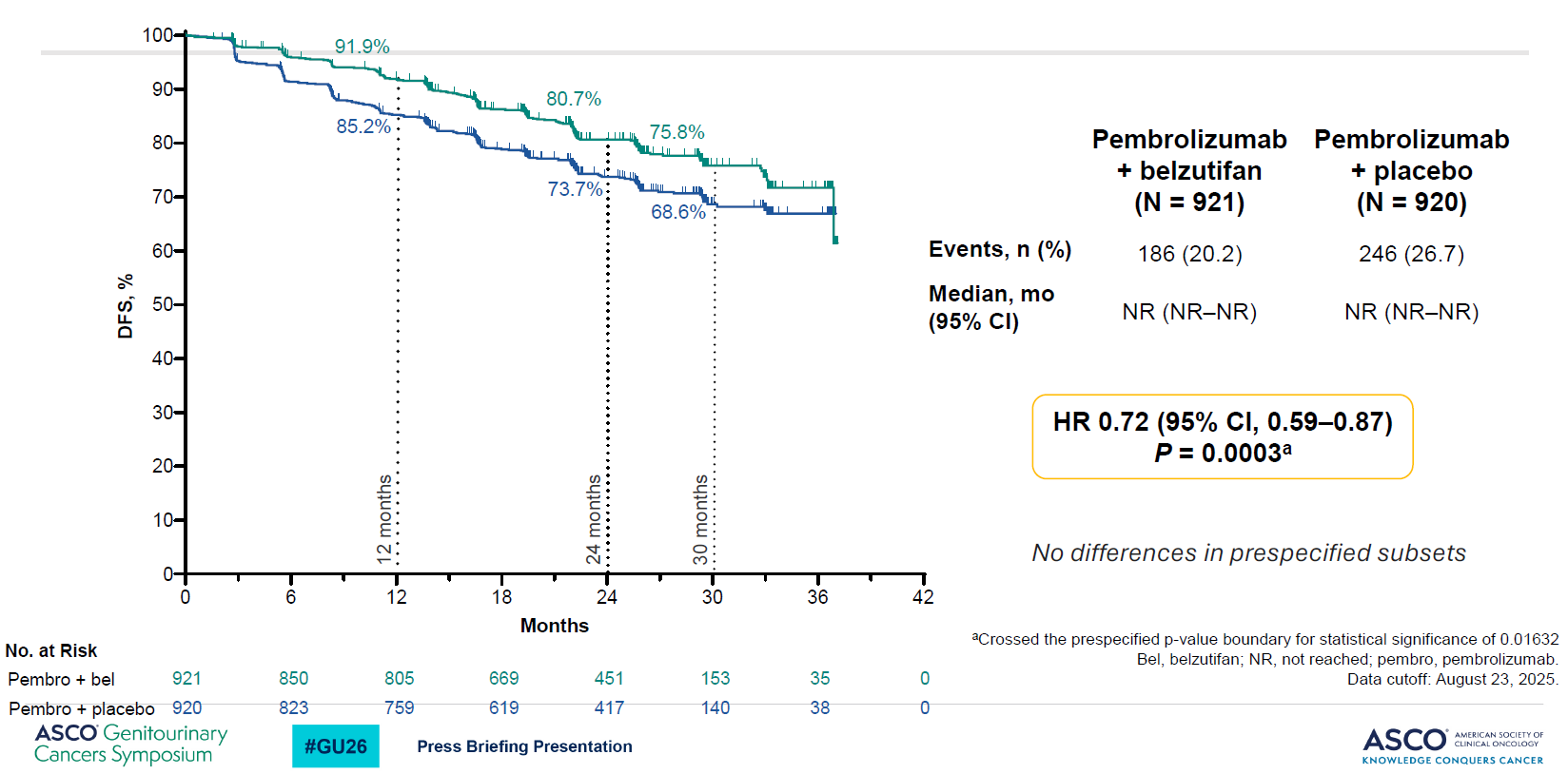
The results of Litespark-022 left a similar impression, with the study, combining Welireg with Keytruda and comparing against Keytruda in adjuvant kidney cancer, yielding a strong result on its primary endpoint of disease-free survival.
There was also a numerical, non-statistically significant OS benefit (hazard ratio of 0.78, p=0.12), which in the case of an adjuvant study like this is already a promising sign. Clearly here there’s the possibility of an OS benefit being hit, given that so far only 87 deaths have occurred, out of 300 needed to trigger final analysis.
On toxicity again anaemia (seen at grade 3 or higher in 12% of Welireg plus Keytruda recipients) and hypoxia (5%) stood out, but also there were increases in liver enzyme elevation with the combo.
One possible question concerns the extent to which the Welierg combo in Litespark-022 was flattered by underperformance of Keytruda control. Landmark 12-month DFS of 85% appears to be in line with the Merck drug’s registrational Keynote-564 study, but the 24-month rate for Keytruda was 78% in Keynote-564 but only 74% in Litespark-022.
Disease-free survival (primary endpoint) in Litespark-022
1323