ASCO-GU – Flare hits an “undruggable” target
But biomarker enrichment and toxicity remain key for FX-909.
But biomarker enrichment and toxicity remain key for FX-909.

The private US biotech Flare Therapeutics has been commended for pursuing PPAR-γ inhibition with its lead project, FX-909. Substantial data from a phase 1 study, however, suggest that the path towards finding a therapeutic window for FX-909 will have to be negotiated carefully, and patients will likely have to be preselected for having high expression of PPAR-γ.
The late-breaking results, presented over the weekend at the ASCO genitourinary cancers symposium, concern a urothelial bladder cancer cohort of a phase 1 solid tumour trial testing four doses. Though a headline response rate of 40% seems impressive it relates to a post-hoc analysis of PPAR-γ-high tumours, and the highest two FX-909 doses are being abandoned.
As an oncology target PPAR-γ is highly unusual, its agonism having been extensively pursued in liver diseases. Presenting the ASCO-GU data Dr Matthew Galsky, of the Icahn School of Medicine at Mount Sinai, said PPAR-γ was the master regulator of the luminal lineage in urotelial cancer, but earlier “antagonists” were unable to inhibit PPAR-γ signalling completely and to repress PPAR-γ transcriptional activity.
FX-909, however, is a PPAR-γ “inverse agonist”, and initial data from the phase 1 study showed a 16% ORR among 31 urothelial carcinoma patients presented at last year’s Triple (AACR-NCI-EORTC) meeting. There were a further four responses, but these weren’t confirmed.
High expressers
At ASCO-GU Galsky zeroed in on patients with high expression of PPAR-γ, identified by a newly developed immunohistochemistry assay. This determined – on a post-hoc basis – that the threshold for FX-909 activity was ≥60% by tumour proportion score.
This subgroup concerned 25 evaluable patients, 10 of whom developed a partial response at any time. However, five of these were unconfirmed, so formally the ORR by Recist criteria stands at 20% – probably still impressive given that patients had to have failed three or more prior therapies, and nearly 80% had received Padcev.
FX-909 activity in urothelial cancer with ≥60% PPAR-γ expression
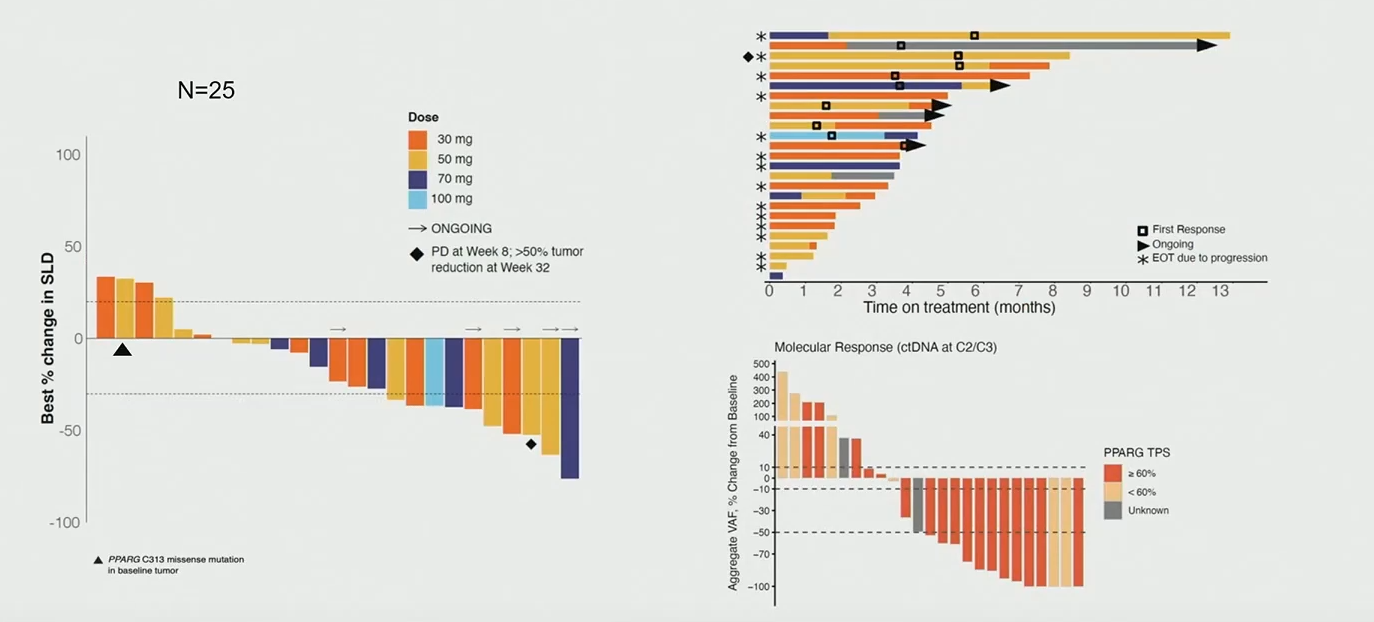
This initial study stage concerned four FX-909 daily doses: 30mg, 50mg, 70mg and 100mg. The highest was associated with two dose-limiting toxicities (hyperglycaemia and proteinuria), Galsky told ASCO-GU, while 70mg had one DLT of anaemia; only 30mg and 50mg are therefore being taken forward into a randomised phase 1b phase.
This doesn’t seem like a problem at present, since initial activity was seen across all doses, and including unconfirmed responses the response rate among the 18 patients given 30mg or 50mg was 39%. But of course these are all post-hoc findings, and Flare’s task now is to show that they hold up prospectively, in the two lowest doses, in patients with ≥60% PPAR-γ expression.
Despite doubts, scoring an ASCO-GU late-breaker is a success for Flare, which raised $123m in a March 2023 series B round, and whose only other disclosed pipeline project is a preclinical AR(on) inhibitor, FX-111. In his presentation Galsky thanked the company for pursuing PPAR-γ, which was “previously considered undruggable”.
1782