
BeOne chases Pfizer in breast cancer

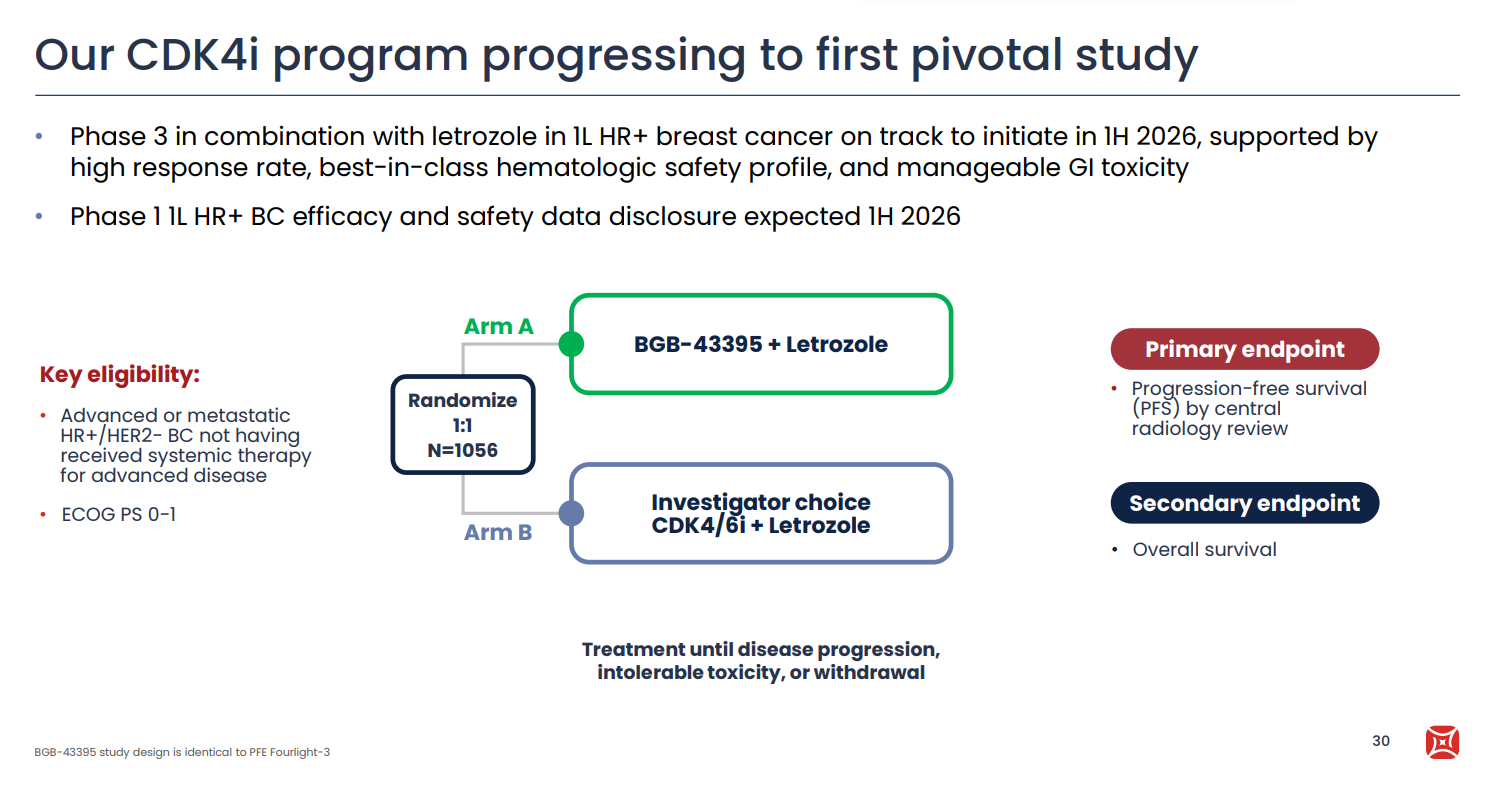
BeOne Medicines has a track record of carrying out head-to-head trials, and it will go down this route again with the phase 3 study of its CDK4 inhibitor BGB-43395. The company last year revealed plans to go pivotal in front-line breast cancer in 2026, but disclosed the first concrete details in its fourth-quarter earnings presentation on Thursday. The as-yet-unnamed trial in ER-positive, HER2-negative disease will test BGB-43395 plus letrozole, versus investigator’s choice of CDK4/6 inhibitor plus letrozole, with a primary endpoint of progression-free survival. CDK4/6 inhibitors like Novartis’s Kisqali and Pfizer’s Ibrance are widely used, but have been linked with neutropenia, which it’s hoped could be reduced by selectively targeting CDK4. The asset to catch here is Pfizer’s atirmociclib, which in January 2025 started the identically designed Fourlight-3 trial, which similarly goes head to head against a CDK4/6 inhibitor in the front line. Both projects have a high bar to clear: Kisqali plus letrozole produced a median PFS of 25 months in its first-line Monaleesa-2 trial. Meanwhile, BeOne last year dropped development of BGB-43395 in second-line breast cancer, citing an evolving competitive landscape.
2575













