
Approval completes Corcept's turnaround

Corcept Therapeutics has pulled off a significant turnaround for its once-troubled glucocorticoid receptor antagonist relacorilant, winning approval in ovarian cancer and sidestepping the toxicity concerns that derailed this drug in Cushing's syndrome. Now branded Lifyorli, relacorilant secured US approval months before its July PDUFA date, backed by survival data from the pivotal Rosella trial, which most recently showed that adding the drug to Abraxane cut the risk of death by 35%. The green light is particularly notable given relacorilant's history: in Cushing's syndrome Corcept was handed a complete response letter after regulators flagged a risk of drug-induced liver injury. This time, however, those concerns appear to have faded, with no liver-related adverse events reported in the Rosella trial at last year's ASCO; the label carries no boxed warning for hepatotoxicity. The approval represents a reversal for relacorilant in oncology following a CRL and 2021 decision to halt phase 3 trials in pancreatic cancer, where the benefit fell short of justifying further study. The company has nevertheless kept a foothold in pancreatic cancer, continuing to test relacorilant in the phase 2 Trident trial, while also advancing its development in endometrial cancer.
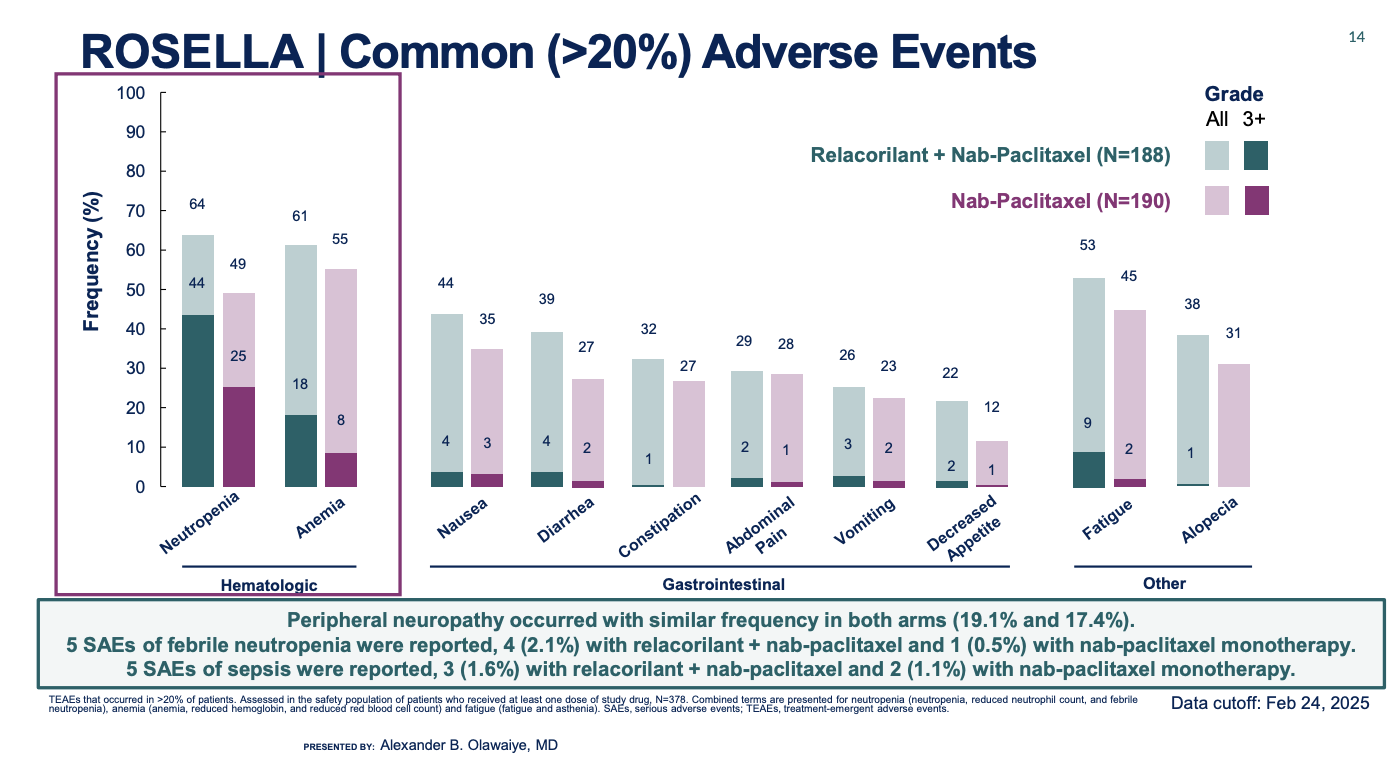
Source: ASCO & Dr Alexander Olawaiye.
Link to OncologyPipeline project
1268













