
Roche takes cevostamab into pivotal trials

Fortified by early multiple myeloma data reported at last year’s ASH conference Roche has taken its anti-FcRH5 T-cell engager cevostamab into pivotal trials. Notably, the phase 3 study, called Cevolution and just listed on the clinicaltrials.gov registry, will test cevostamab in patients who have received as few as one prior therapy, a setting in which the phase 1 Camma-1 study yielded ORRs of 86% and 88% for respective 70mg and 105mg doses, combined with Pomalyst and dexamethasone, as reported at ASH. The conference also saw the first results of Camma-3, in third to 12th-line patients, where the ORR was 39% in all 52 subjects, and 25% in the 26 who had been exposed to anti-BCMA therapy. One thing to watch is toxicity: in Camma-1 there was one death, at 105mg, due to intracranial haemorrhage, while in Camma-3 there were two, both due to infections, though none of the three was considered related to cevostamab. It’s not clear what cevostamab dose is being taken into Cevolution, which will test a Pom-dex combo versus standard of care. OncologyPipeline reveals just two other clinical-stage anti-FcRH5 projects in development: Moderna’s RNA-encoded T-ell engager mRNA-2808, and Legend’s Car-T therapy LCAR-F33S.
Cevostamab + Pom-dex in 2nd-line plus multiple myeloma, Camma-1 trial
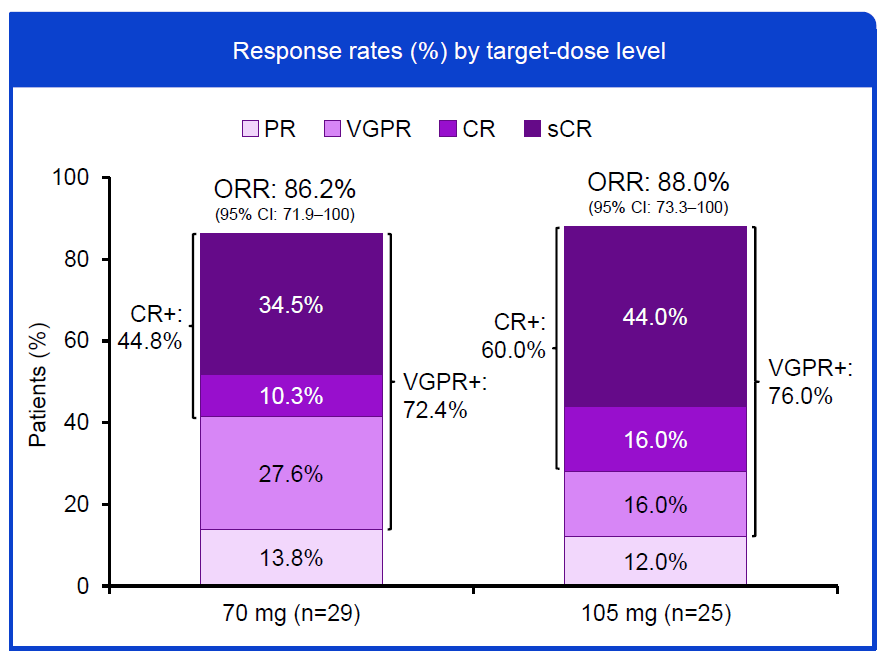
Note: data cutoff 6 Sep 2025. Source: Dr Caroline Hasselbalch Riley & ASH.
Link to OncologyPipeline project
46













