
An unexpected Serena knockback for Astra
A US advisory committee votes against camizestrant, but does endorse a new use for Truqap.
A US advisory committee votes against camizestrant, but does endorse a new use for Truqap.

AstraZeneca’s ingenious plan to have its oral SERD camizestrant approved in an early breast cancer setting has received a serious blow, with a US advisory committee on Thursday voting six to three against the supporting Serena-6 study.
The double adcom was an all-Astra affair, with its afternoon session considering Truqap’s Capitello-281 study in hormone-sensitive prostate cancer – a use that, perhaps also unexpectedly, received a strong endorsement. It now falls to the FDA whether to approve each drug, but it would take some doing for the agency to go against camizestrant’s 6-3 knockback.
Serena-6 enrolled first-line ER-positive, HER2-negative breast cancer patients who had received CDK4/6 and aromatase inhibitors, and monitored them for development of an ESR1 mutation. Patients in whom ESR1m was detected – but who hadn’t had disease progression by imaging – were then given camizestrant plus a CDK4/6 inhibitor, or continued on their original therapy.
Detection not established
However, this treatment setting relies on a shift in medical practice, introducing routine ESR1m testing using ctDNA analysis, and giving a new drug before actual disease progression.
And this was one aspect with which the panel members were uncomfortable; the FDA reviewer said ESR1 detection wasn’t established, though surely this was the point. Ingrid Mayer, an Astra vice-president, told the panel: “A push towards personalised medicine requires rethinking practice.”
Serena-6 was also criticised for not evaluating whether switching at ESR1 detection was better than switching at standard radiographic progression. Early switch necessitated patients forgoing current treatment, from which they might still be benefiting, it was suggested.
A bigger problem was the lack of long-term outcomes data, and the fact Serena-6 hadn’t shown an OS benefit – and indeed might never show one given early survival signals and the fact the trial was “somewhat underpowered”.
Serena-6 survival curves at 58% information fraction
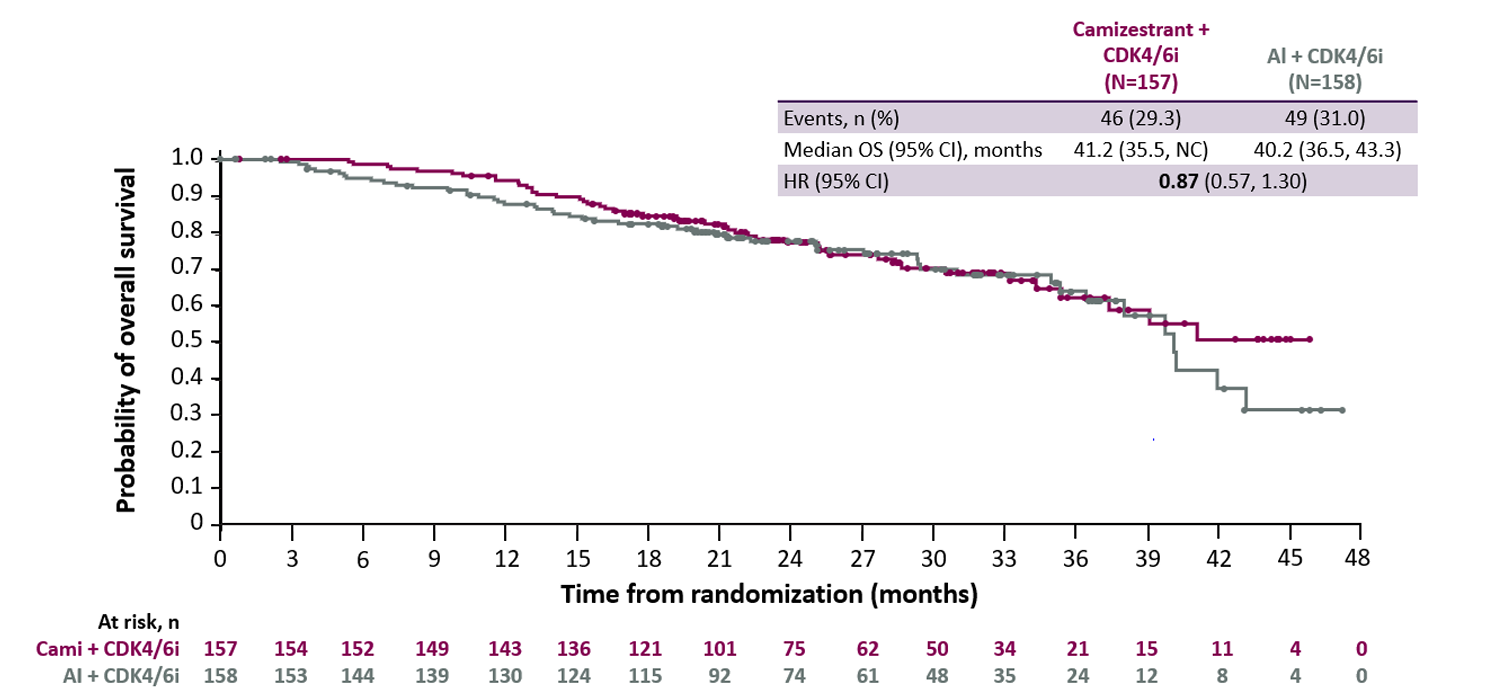
Note: data cutoff 2 Jan 2026. Source: adcom documents.
At ASCO Astra attempted to pre-empt the OS problem by touting data on PFS2, which it claimed was a more relevant real-world endpoint. The FDA reviewer dismissed this, stating: “PFS2 is not adequate to demonstrate clinical benefit” because it doesn’t isolate the effect of the experimental drug.
And there was a cardiac toxicity issue too, with a case of torsades de pointes in a patient given camizestrant plus Novartis’s Kisqali. Astra put this down to Kisqali, but with just 23 of 156 camizestrant patients in Serena-6 receiving Kisqali, rather than another CDH4/6 inhibitor, the safety data on this combo was described as limited.
Capitello OK
The case against Truqap, an AKT inhibitor already approved for PIK3CA/AKT1/PTEN-altered breast cancer, seemed far simpler, and perhaps more damning. Astra is seeking to extend this drug’s use into PTEN-deficient metastatic hormone-sensitive prostate cancer based on the Capitello-281 study, which compared Truqap plus Zytiga versus Zytiga, and whose results were presented at last year’s ESMO.
The agency said it had told Astra that PFS was an acceptable endpoint here, but would have to be supported by an “improvement in OS ... in the absence of a large magnitude of improvement in PFS”. In the event median PFS came in at 33 versus 26 months, and the resulting hazard ratio of 0.81 featured wide confidence intervals, the upper bound of which came close to 1.00, at 0.98.
If that effect appeared modest, OS drew an absolute blank, with a numerical 10% reduction in risk of death yielding a highly non-significant p value of 0.401. In addition to this Astra fielded numerous questions from the adcom panellist Dr Toni Choueiri, of Dana-Farber, as to what was the appropriate IHC cutoff for the PTEN biomarker.
Despite these doubts the adcom voted seven to one that the benefit of Truqap outweighed its risk in Capitello-281's proposed indication.
Capitello-281 survival curves at 26% maturity
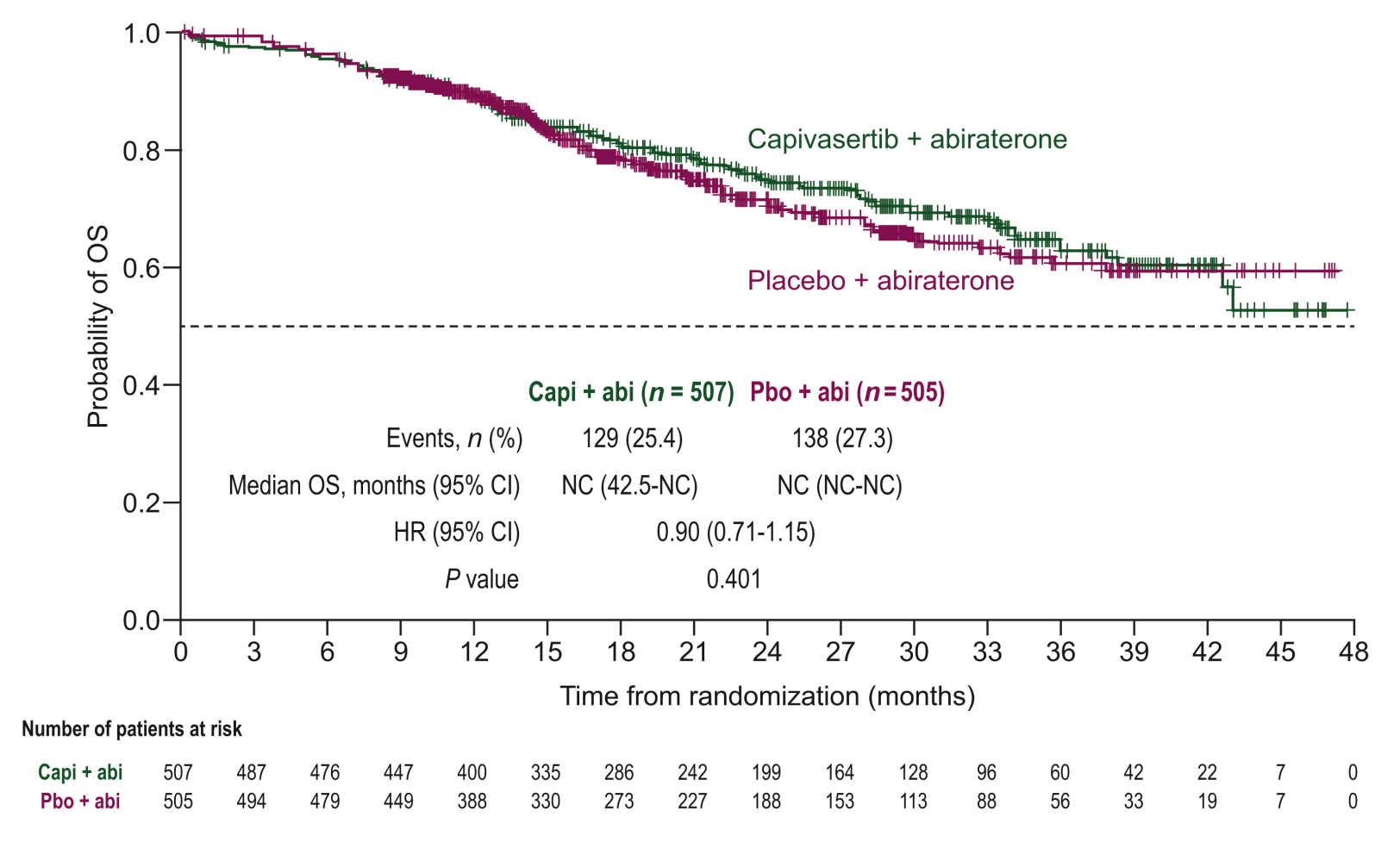
Source: ESMO.
Link to OncologyPipeline project
18













