AACR 2026 – Circle’s focus narrows
First-in-human results prompt a biomarker-defined approach for CID-078.
First-in-human results prompt a biomarker-defined approach for CID-078.

The private US biotech Circle Pharma has reported the first case reports suggesting that its lead project, CID-078, might have a future in a target previously thought to be “undruggable”.
However, the early data Circle has seen have prompted the company to zero in on a more tightly defined patient set, namely those with mutations in the tumour suppressor gene RB1. This could be key to dialling up efficacy for an approach that’s generated sufficient preclinical promise to attract $90m in a 2024 series D financing.
That happened at roughly the same time as CID-078 entered its first-in-human solid tumour trial. It’s this study that featured at a plenary session at AACR on Sunday, with the findings amounting to the first clinical data publicly reported for this molecule.
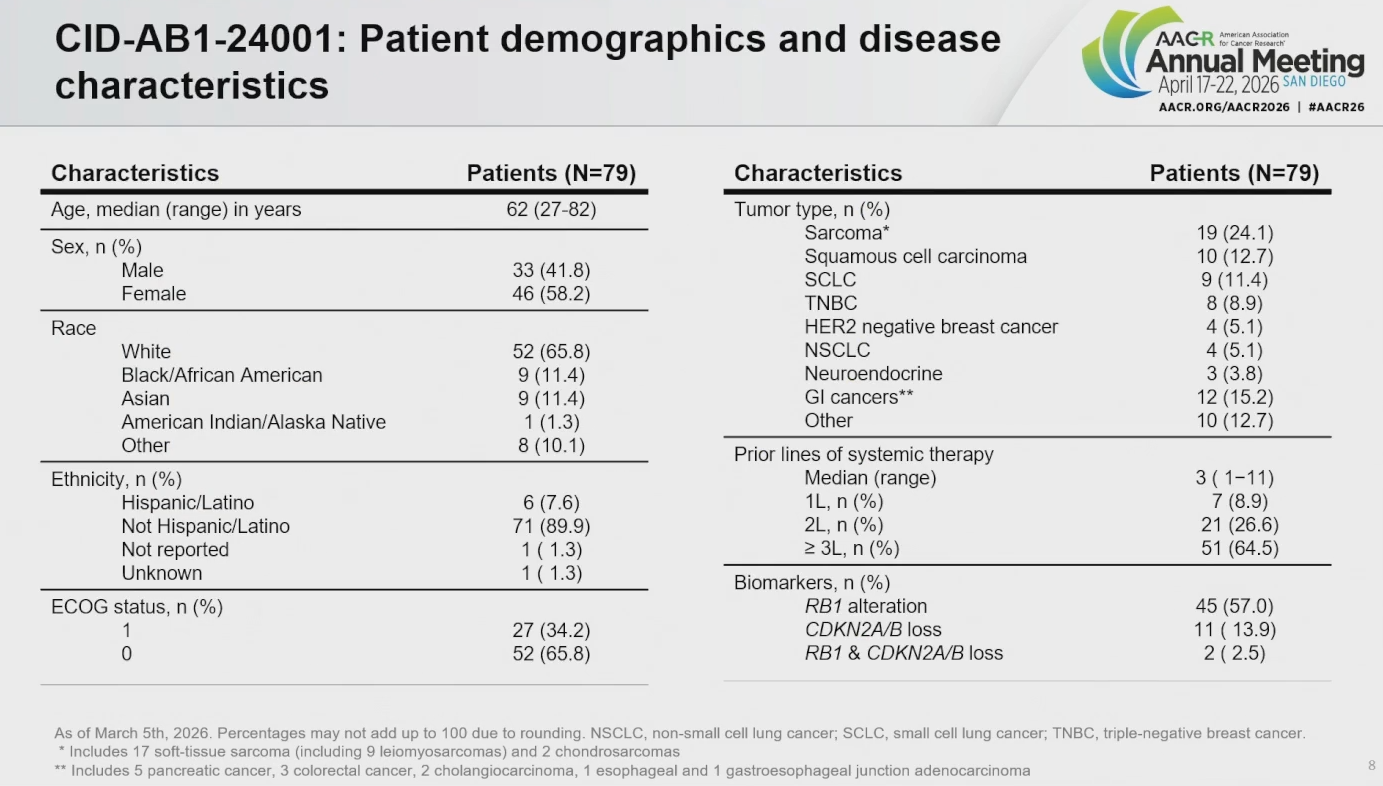
The presentation revealed that 79 patients have now been treated in dose escalation, which recruited heavily pretreated patients with solid tumours not limited to any particular biomarker.
However, this is now changing, with backfill cohorts set to enrol patients whose tumours show RB1 alteration. And future expansion cohorts are planned that will evaluate RB1-driven tumour types such as sarcoma, neuroendocrine carcinoma including SCLC, and triple-negative breast cancer, said Dr Afshin Dowlati of Case Western Reserve University.
Mechanistic rationale
The reason for is linked to the mechanism of CID-078, an A/B RxL inhibitor that binds to a “previously undruggable” hydrophobic patch on cyclin A and B complexes. The aim of this is to overcome cell cycle dysregulation caused specifically by loss of RB1; such loss occurs in 80-90% of SCLCs and leiomyosarcomas, Dowlati told AACR.
Still, backing for CID-078’s activity is limited to two highlighted confirmed responses: one in a seventh-line RB1-altered pleomorphic sarcoma patient, and one in a fourth-line subject with neuroendocrine tumour. These concerned 480mg and 360mg twice-daily doses – around the middle of the 40-720mg range tested in this escalation phase.
The good news is that Circle apparently has scope to dose higher, as Dowlani said no relationship had been seen so far between increased dosing and toxicity. Adverse events, mostly grade 1 or 2, included a 13% rate of treatment-related ALT elevation, with two grade 3 ALT increases seen at 600mg and in a 300mg backfill cohort.
Assuming that such events remain manageable Circle could look to higher CID-078 dosing, which in a biomarker-defined population could see efficacy rise.
CID-078 safety in phase 1 dose escalation
Souce: Dr Afshin Dowlati & AACR.
Link to OncologyPipeline project
182