Allogene finds a way forward for allogeneic Car-T
After setbacks, cema-cel might have a path to approval in lymphoma.
After setbacks, cema-cel might have a path to approval in lymphoma.

In a lymphoma landscape squeezed by second-line autologous Car-T therapy on the one hand, and by bispecific T-cell engagers on the other, Allogene hopes to have found a niche that’s now been backed by the first data from the Alpha-3 trial of its allogeneic anti-CD19 Car-T project cemacabtagene ansegedleucel.
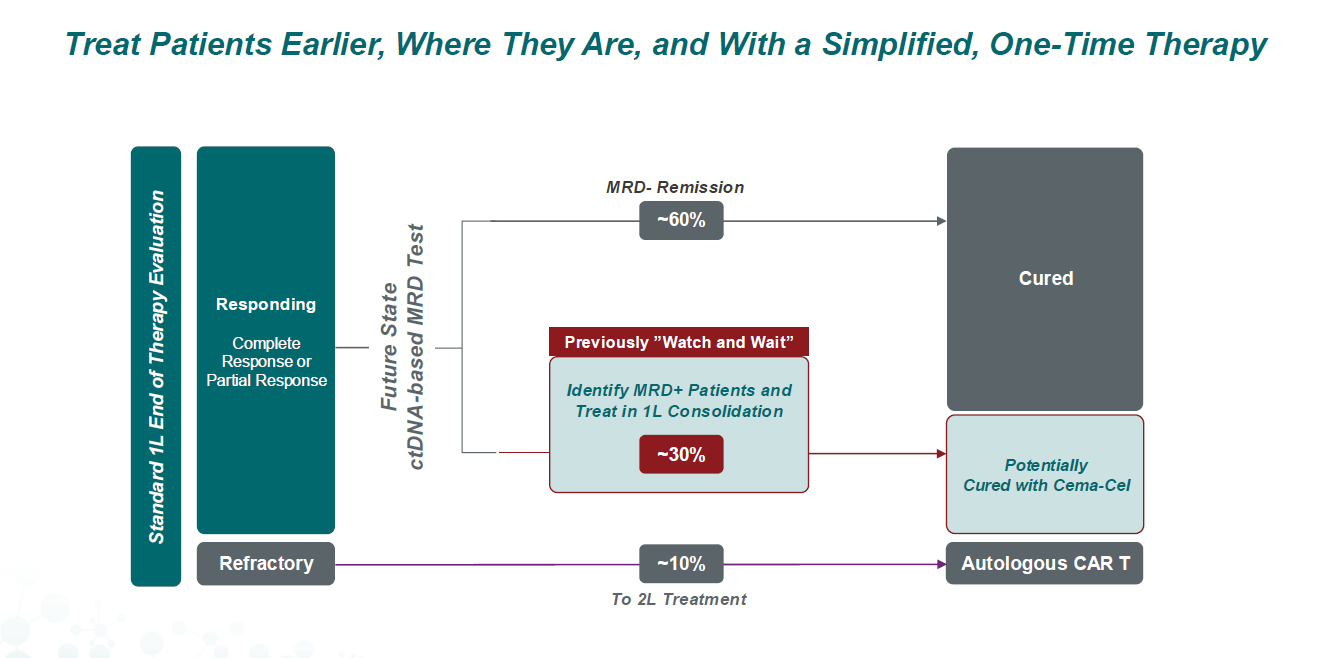
The study tests cema-cel as consolidation in diffuse large B-cell lymphoma patients who are MRD-positive but are still in response to first-line R-Chop therapy. On Monday Allogene reported an interim futility analysis from Alpha-3’s fist 24 patients, showing that 58% of cema-cel subjects became MRD-negative, versus 17% of those who underwent observation alone.
This gives some backing in a very small dataset to Allogene’s contention that mere observation – the current standard of care for DLBCL patients in response to front-line chemo – isn’t enough. Now the company must repeat the success in the entire 220-strong Alpha-3 cohort, and hope that it translates into a hit on the study’s primary endpoint of event-free survival.
Should that result come in positive it would support US filing, and mark an important change of fortune for Allogene, which along with other allogeneic Car-T players has found the going tough. If cema-cel comes to market its commercial success would then hinge on physicians implementing ctDNA testing, to determine responding patients’ MRD status, into standard practice.
A possible new treatment paradigm for DLBCL patients in response to 1st-line R-Chop
For now the signs are good, with Alpha-3’s futility analysis coming in almost exactly as Allogene had expected. The group set the goalposts for this readout, saying it expected around two of 12 control patients to undergo “spontaneous” switch to MRD-negative, versus six or so for those treated with cema-cel, resulting in a 25 to 30 percentage point absolute delta.
In the event MRD clearance was seen in seven cema-cel patients, and in two of those on observation alone, resulting in a 41.6-point difference.
Equally important, given cema-cel’s death scare last August, was the therapy’s relative lack of toxicity; there was no cytokine release, ICANS or graft-versus-host disease, at any grade, in the cema-cel cohort, and no treatment-related deaths. Infections and other neurological events were all described as low grade.
This supports Allogene’s claim at the time that the earlier death was related not to cema-cel but to enhanced lymphodepletion using the anti-CD52 antibody ALLO-647. Alpha-3 originally tested cema-cel with standard or ALLO-647 lymphodepletion, but after the death all ALLO-647 cohorts were axed, and the futility analysis relates only to patients given standard lymphodepletion.
Alpha-3 has been enrolling relatively slowly, and Allogene expects to complete recruitment of all 220 patients by the end of next year. Before then, in mid-2027, the trial will undergo its first interim analysis for EFS – a key event given that the relationship between MRD clearance and EFS isn’t “linear”, according to Allogene.
Primary analysis would then occur in mid-2028, with the data supporting a US filing. The company ended last year with $258m in the bank; that was expected to last into the first quarter of 2028, but with Allogene’s shares opening up 45% on Monday the group now has scope to raise more.
222