Ideaya’s darovasertib plan comes together
Optimum-02 virtually replicates phase 1, and a filing will follow.
Optimum-02 virtually replicates phase 1, and a filing will follow.

The registrational phase 2/3 Optimum-02 study of Ideaya’s pan-PKC inhibitor darovasertib in uveal melanoma has largely replicated earlier mid-stage findings, but this time against an active comparator. And, with statistical significance hit on progression-free survival, darovasertib is headed for a US accelerated approval filing in the second half of this year.
The results suggest that Servier was right to license ex-US rights to darovasertib last year, in a deal that brought Ideaya a $210m up-front windfall. If the data lead to approval this would complete a remarkable journey for darovasertib, which was originated by Novartis before that company sold it to Ideaya in 2018 for a mere $6m.
Ideaya embarked on Optimum-02 in 2023, testing darovasertib plus Xalkori in first-line metastatic uveal melanoma in patients who do not carry the HLA-A2 serotype. This population lies outside the scope of Immunocore’s HLA-directed T-cell engager Kimmtrak, which is approved for uveal melanoma patients who are HLA-A*02:01-positive; these make up nearly 50% of the Caucasian population.
Ideaya’s enthusiasm was driven by the phase 1/2 Optimum-01 trial, where darovasertib plus Xalkori scored median PFS of 7.1 months; that was said to compare favourably against Opdivo plus Yervoy’s historical three-month mPFS, though of course Ideaya still had to demonstrate such a benefit prospectively.
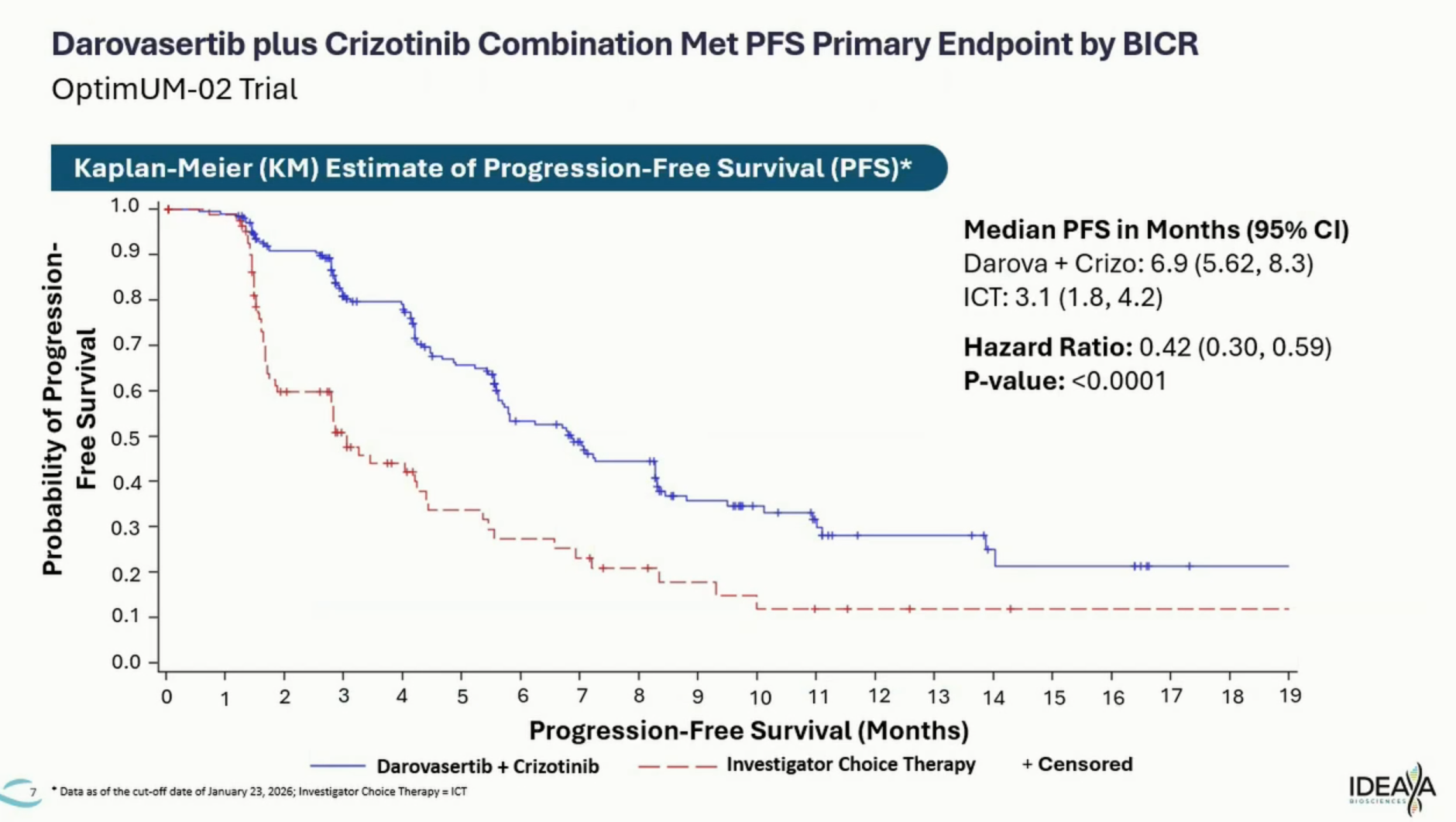
This is precisely what it has now done, with the figures coming in remarkably close to those earlier data. On Monday Ideaya said darovasertib/Xalkori scored 6.9 months of mPFS, versus 3.1 months for investigator’s choice (Keytruda monotherapy or Opdivo plus Yervoy).
This resulted in a strong reduction in risk of progression or death of 58% (p<0.0001). Overall survival from Optimum-02, expected to serve to confirm a potential accelerated approval, was said to be showing an “early trend in improvement” at 10 months’ median follow-up. Ideaya shares opened up 18%.
PFS curves from Optimum-02
Though the focus for the readout is PFS, these data were supported by an overall response rate benefit – 37% for the combo versus 6% for investigator’s choice. The fact five responses to the darovasertib combo were deemed complete by blinded independent central review, versus none in the control cohort, came as a pleasant surprise.
While darovasertib’s safety profile can’t be scrutinised until Optimum-02 data are presented in full at a medical meeting, with no specific worries disclosed at present the Xalkori combo appears to be approvable.
In comparison, in the HLA-A*02:01-positive space Kimmtrak was approved on the back of median OS of 21.7 months, versus 16.0 months for Keytruda, Yervoy or decarbazine chemo (0.51 hazard ratio), in a trial that showed a modest mPFS benefit (3.3 versus 2.9 months, HR=0.73).
Uveal melanoma is a relatively niche setting, with Ideaya citing 3,000 US patients diagnosed a year, half of whom progress to metastatic disease. The company wants to use a cohort of HLA-A*02:01-positive patients from Optimum-01 to back compendia listing, and is separately testing darovasertib monotherapy in neoadjuvant (Optimum-10) and a Xalkori combo in adjuvant (Optimum-11) settings.
It’s notable that Ideaya sees darovasertib as just its initial route to market, recently dosing the first patients in phase 1 trials of the KAT6/7 inhibitor IDE574, PRMT5 inhibitor IDE892, anti-B7-H3 x PTK7 ADC IDE034, and anti-DLL3 ADC IDE849. Its MAT2A inhibitor IDE397 is also worth watching.
The group ended last year with over $1bn in the bank, and it doesn’t look likely to rest on its laurels any time soon.
151