
Time and cash are running out for Innate

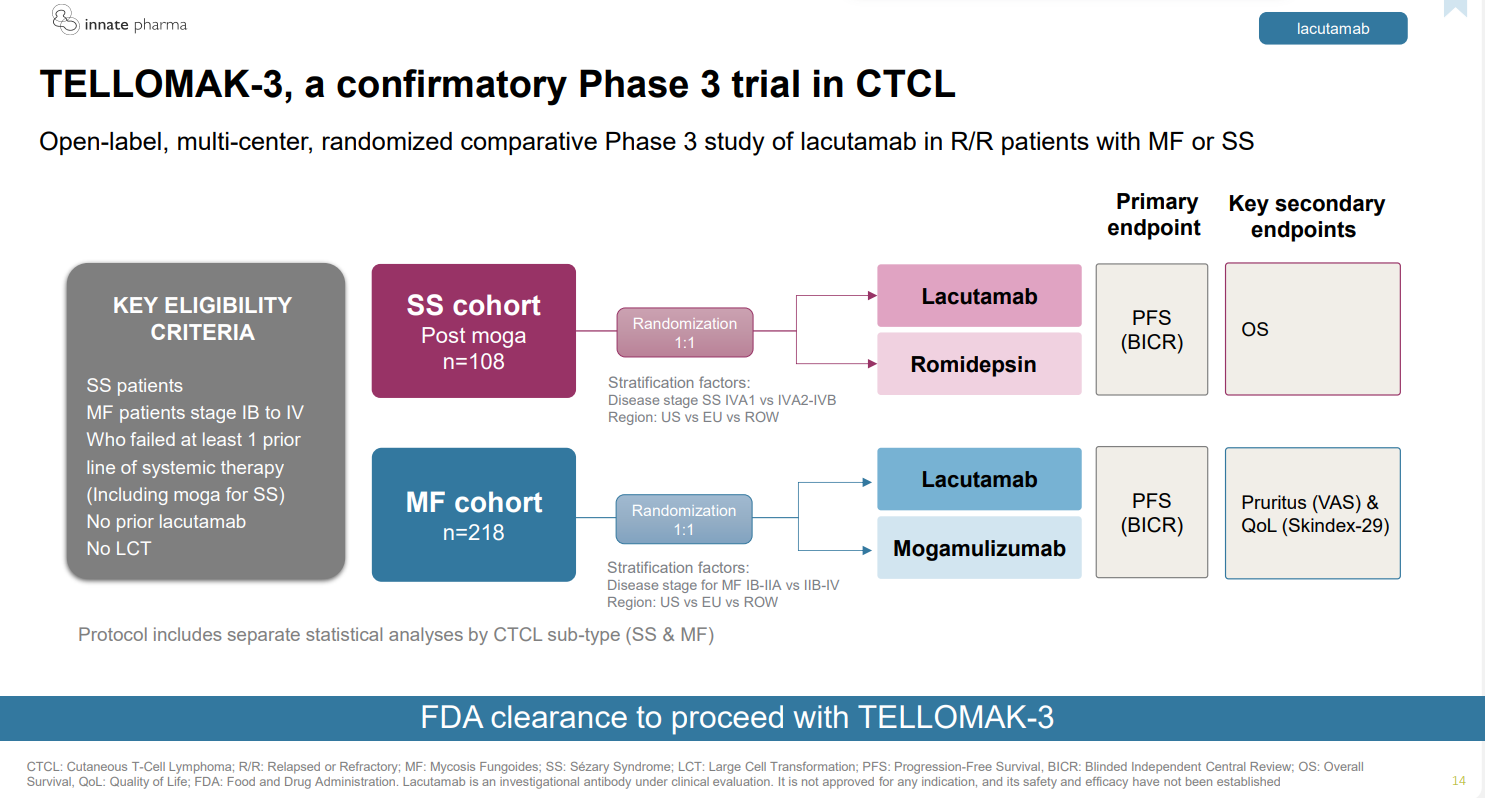
Innate Pharma’s NK-cell engagers have had a torrid time recently, and the latest one heading for the scrapheap is IPH6501, a wholly owned CD20-targeting asset. Innate said in its fourth-quarter earnings release that it had discontinued this after a phase 1/2 trial in B-cell non-Hodgkin’s lymphoma. Data, once expected last year, will be presented in 2026, but the writing had been on the wall since the group disclosed “limited signs of activity” last September. Sanofi has also walked away from several Innate NK projects including, most recently, the BCMA-targeting IPH640; however, focus had already shifted to autoimmune disease. Separately, Innate is facing a delay to its wholly owned lead project, the anti-KIR3DL2 antibody lacutamab; the phase 3 Tellomak-3 trial is now slated to start in the second half, versus the first half previously, but even this timeline looks uncertain as it depends on fund-raising. Innate has €44.8m in cash, enough to last only until the third quarter. Tellomak-3 will encompass two cutaneous T-cell lymphomas: Sézary syndrome and mycosis fungoides. Innate hopes to file for accelerated approval in the former, based on the phase 2 Tellomak trial – but this depends on Tellomak-3 being under way.
667













