
AACR 2026 – Matisse pains a mixed picture for Innate
IPH5201 plus Imfinzi in perioperative NSCLC looks better than some but not other historical studies.
IPH5201 plus Imfinzi in perioperative NSCLC looks better than some but not other historical studies.

Innate Pharma’s AstraZeneca-partnered anti-CD39 MAb IPH5201 has shown limited promise in a mid-stage uncontrolled trial in lung cancer, and the jury is still out on just how positive these are.
Interim results with IPH5201 plus Astra’s anti-PD-L1 MAb Imfinzi and chemo, presented during an AACR plenary session on Tuesday, look better than historical data with Imfinzi plus chemo alone, particularly in PD-L1-high patients.
However, in a cross-trial comparison against Bristol Myers Squibb’s Opdivo plus chemo, the latest results seem less impressive.
More colour
Matisse aims to enrol 70 patients with resectable, stage II-IIIA NSCLC, regardless of PD-L1 expression. The interim analysis presented at AACR involves the first 40 patients.
Subjects received neoadjuvant IPH5201 plus Imfinzi and chemo, then adjuvant IPH5201 plus Imfinzi. The primary efficacy endpoint is pathologic complete response.
At a cutoff date of 11 December 2025, the pCR was 28% across the whole patient population. Among patients with PD-L1 expressions of <1%, ≥1% and ≥50%, pCR was 8%, 36% and 50% respectively.
The AACR discussant, MD Anderson’s Dr Tina Cascone, concluded that there was “clearly” a signal in the PD-L1-high population. The big question, though, is whether the IPH5201 adds anything to checkpoint blockade plus chemo alone.
Cross-trial comparisons
Cascone highlighted a cross-trial comparison, first against Astra’s Aegean trial of Imfinzi plus chemo, which produced a pCR of 17% across the entire population, and 28% in PD-L1-high patients. The comparison therefore favoured Matisse, although Cascone urged caution, especially given that the Matisse data involved 40 patients, versus 740 in Aegean.
However, bringing Opdivo/chemo trials into the mix muddied the waters, with Checkmate-77T and Checkmate-816 both producing similar results to Matisse.
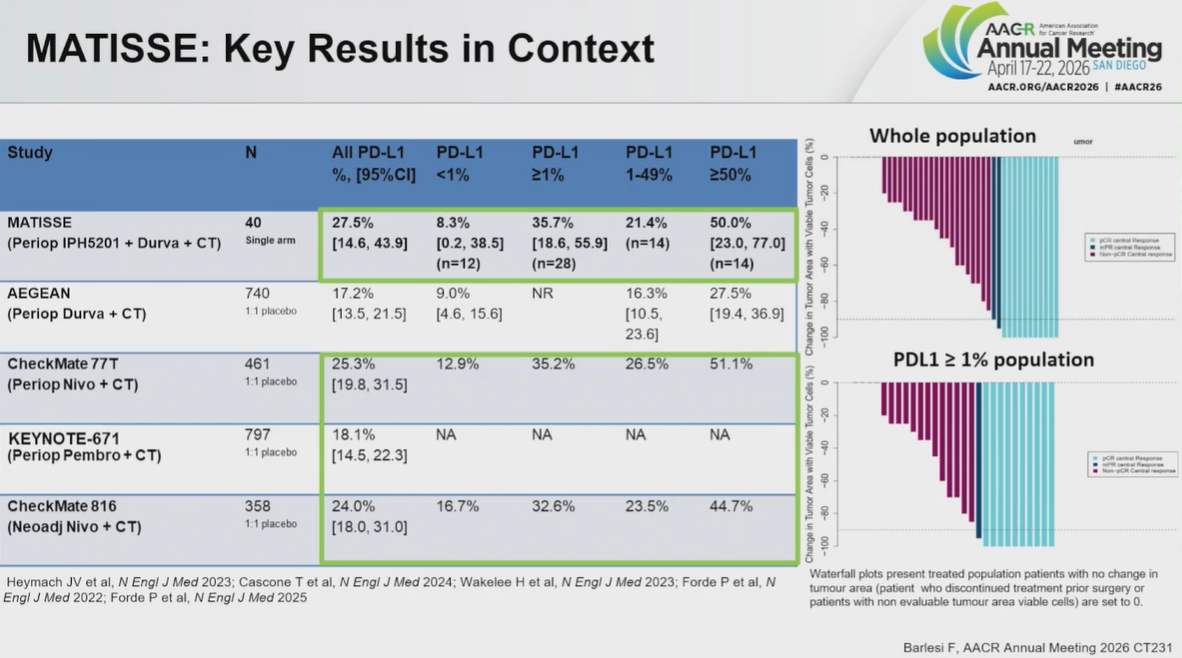
Cascone also noted that Matisse was designed with a target pCR of 40%. It’s possible for results to improve at the final analysis, although they might also worsen. The trial is continuing to recruit patients with ≥1% PD-L1 expression.
The latest release might not do much to alleviate nerves about Innate’s dwindling cash pile, although the company’s chief executive, Yannis Morel, told ApexOnco ahead of AACR that it wasn't not investing heavily in IPH5201.
Astra began co-developing the project in 2018, building on a 2015 alliance over what’s now one of Innate’s “high-value assets”, the anti-NKG2A MAb monalizumab.
117













