
Revolution’s big reveal approaches
The group’s first pivotal readout, in second-line pancreatic cancer, is coming.
The group’s first pivotal readout, in second-line pancreatic cancer, is coming.

Revolution Medicines has built a $20bn market cap on the promise of improved RAS inhibition, and soon it’ll have a chance to put its claims to the test, with the first pivotal data on its pan-RAS contender daraxonrasib.
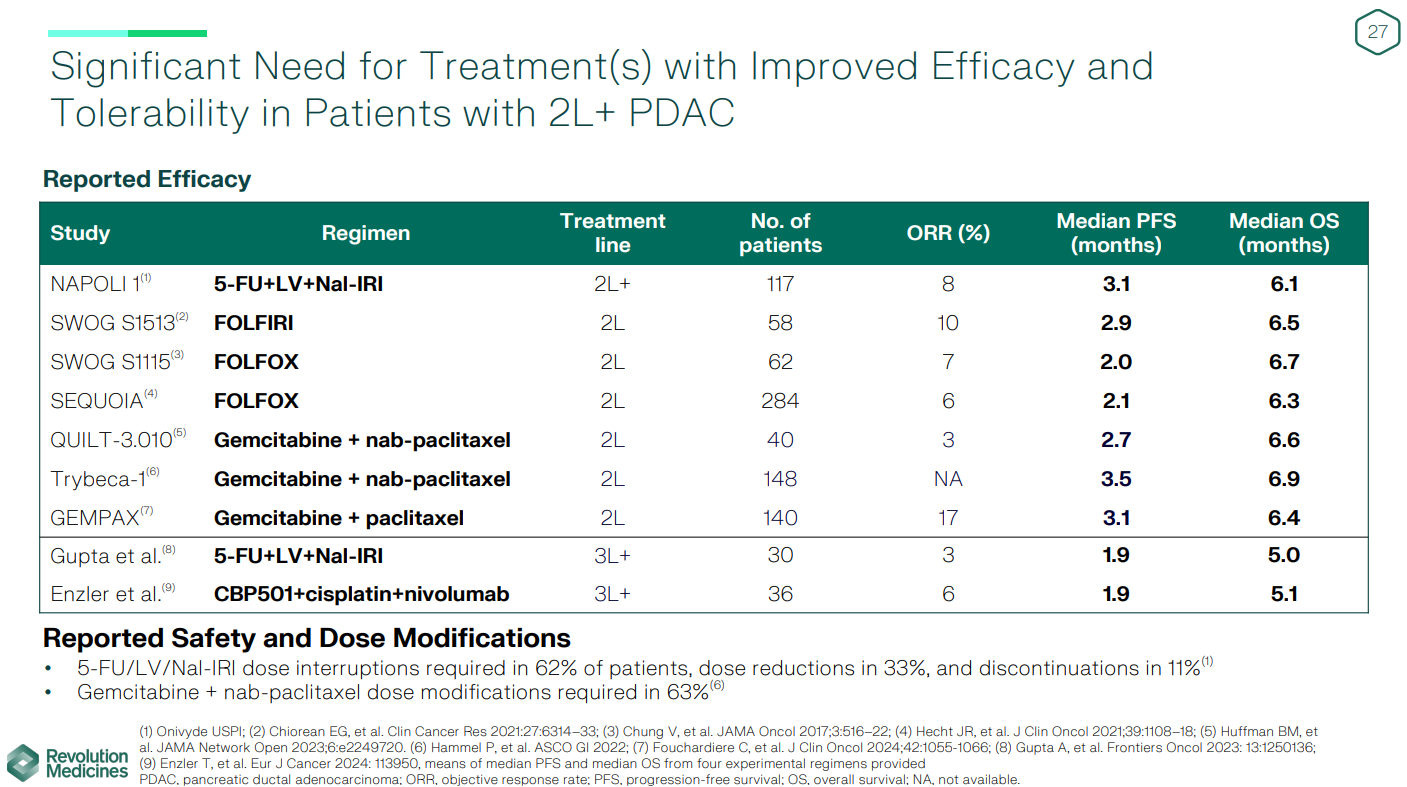
The Rasolute-302 trial in second-line pancreatic cancer is set to read out this half, and the bar to clear looks relatively low. The standard of care here is chemotherapy, which has historically produced median progression-free and overall survival of around 3 months and 6-7 months respectively.
Daraxonrasib is designed to target GTP-bound (active) form of RAS, also known as its “on” state, while currently approved KRAS G12C inhibitors such as Amgen’s Lumakras and Bristol Myers Squibb’s Krazati target the “off” state. It’s hoped that “on” inhibitors such as daraxonrasib could address the resistance seen with the incumbent drugs.
So far, daraxonrasib has produced promising early results, although some of Revolution’s updates have been confusing, and there have been high rates of rash and gastrointestinal side effects.
Rasolute
These will be watched in Rasolute-302, which tests daraxonrasib at 300mg, versus investigator’s choice of chemo, in around 500 pancreatic ductal adenocarcinoma (PDAC) patients who have previously received one line of chemo.
Interestingly, the study enrols RAS-mutant and wild-type patients alike, although over 90% of PDAC patients have RAS mutations, Revolution estimates. And the co-primary endpoints, PFS and OS, will only be assessed in the RAS G12-mutant population, while PFS and OS in all-comers are among the secondary endpoints.
Revolution has given an indication of what to expect in the control arm, citing various studies that have produced median PFS of 2.0-3.5 months and median OS of 6.1-6.9 months with chemo.
In phase 1, daraxonrasib far outstripped these historical numbers, with median PFS of 8.5 months and OS of 13.1 months among second-line G12-mutant patients receiving a 300mg dose. Still, this analysis only included 26 patients, and numbers tend to dwindle in phase 3.
Evercore ISI’s Cory Kasimov cited a median PFS of 5-6 months and median OS of 10-11 months as the bar for daraxonrasib to hit; he added that the unmet need in this indication was so severe that even a “modest gain versus chemo [would be] practice changing”.
However, adverse events could also play a part. In the phase 1 trial rash was seen in 90% of second-line-plus patients (7% at grade 3 or higher), while 52% experienced diarrhoea (4% at grade 3 or higher).
Kasimov suggested that, in the real world, rash and GI events could be managed with prophylactic measures and dosing holds or reductions. He estimated that, if daraxonrasib made it to market, it could be priced at a 20% premium to approved RAS drugs.
He added that daraxonrasib could become “one of the strongest oncology launches ever”, and predicted $7.4bn in unadjusted peak sales in PDAC, well above analysts' consensus of $5.3bn.
Still, speculation that Revolution could be acquired, first by AbbVie then Merck & Co, has so far come to nothing, suggesting that the biotech could be overvalued, especially based on results from small, uncontrolled studies.
Rasolute-302 will need to be a knockout if Revolution is to live up to its hype.
Phase 1/2 data with daraxonrasib 300mg in second-line pancreatic cancer
| RAS G12 mutant | RAS mutant | |
|---|---|---|
| N | 26 | 38 |
| cORR | 35% (9/26) | 29% (11/38) |
| mPFS | 8.5mths | 8.1mths |
| mOS | 13.1mths | 15.6mths |
Note: data cutoff 30 June 2025. Source: company releases.
3892













