AACR 2026 – Revolution’s second project impresses again

Revolution Medicines is riding high on pivotal data with its pan-RAS inhibitor daraxonrasib, but its KRAS G12D-selective inhibitor zoldonrasib also continues to look good. New data from a phase 1 study, presented at an AACR plenary session on Sunday, found that zoldonrasib at 1.2g led to a confirmed response rate of 52% among 27 patients with previously treated G12D-mutant NSCLC, and median progression-free survival of 11.1 months (median overall survival wasn’t reached). On the face of it the response rate has waned from the 61% ORR the company claimed at AACR last year among 18 patients; however, that number included unconfirmed responses. It also encompassed NSCLC patients who had received various prior therapies, while the latest release focused on the tough-to-treat post-PD-(L)1 population. Side effects have ticked up, though, with 13% of patients experiencing a grade 3 treatment-related adverse event, and 5% discontinuing owing to an adverse event (versus 2% and 1% respectively previously, across tumour types). Still, there continue to be no grade 4 or 5 adverse events. Revolution plans to start a pivotal study of a zoldonrasib combo in first-line NSCLC, Rasolve-308, this half, although it hasn’t disclosed details. Other pivotal G12D-selective projects include Astellas’s degrader setidegrasib.
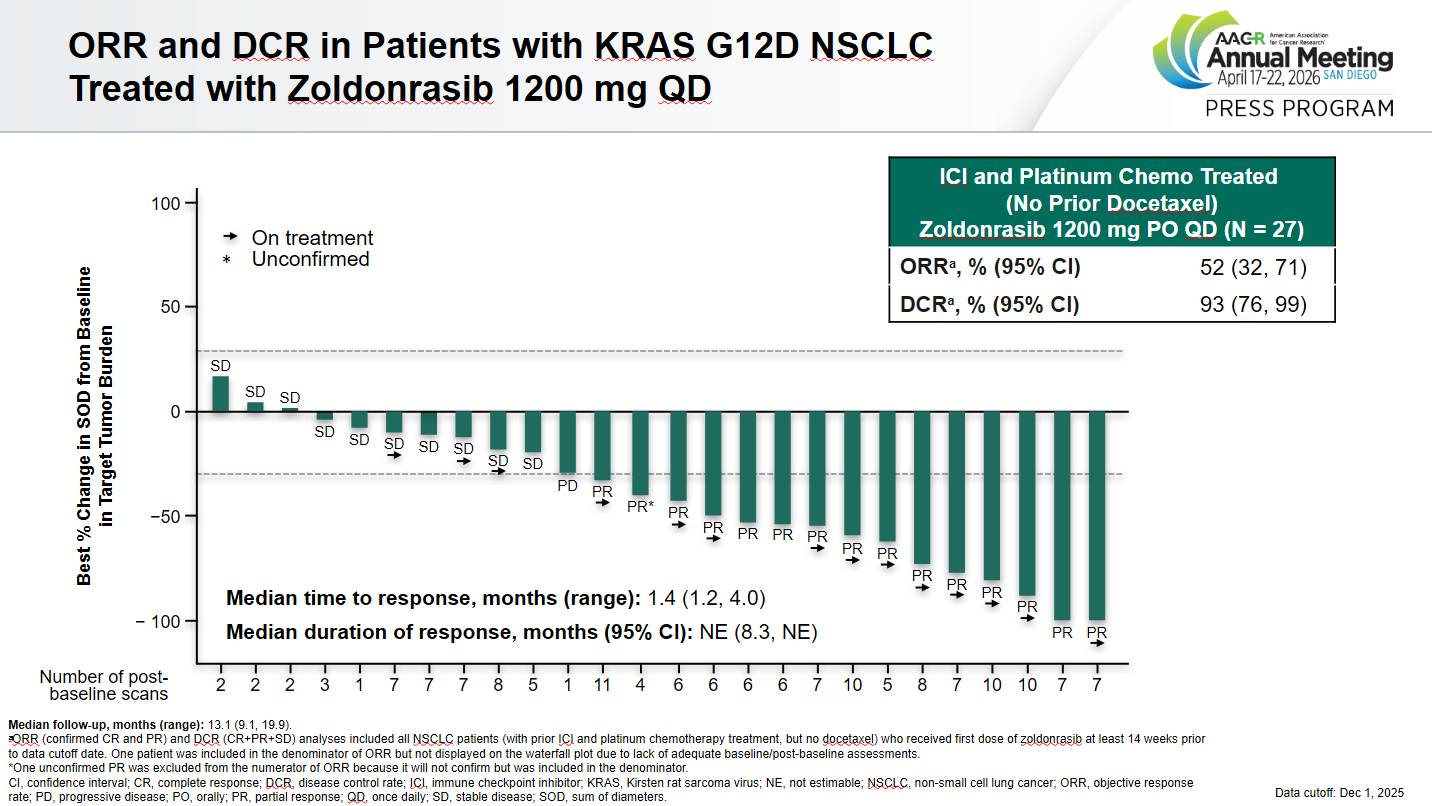
Source: Dr Jonathan Riess & AACR
Link to OncologyPipeline project
278