
ESMO 2023 – not so fast, Welireg
The Merck & Co drug's first successful phase 3 study might not be all it’s cracked up to be.
The Merck & Co drug's first successful phase 3 study might not be all it’s cracked up to be.

After the euphoria over Welireg’s first successful phase 3 trial, today it was time for the reckoning. Revelation of the Litespark-005 study’s survival curves in an ESMO late-breaker brought very cautious praise from the discussant, and could cloud the drug’s ability to break into the broad renal cancer setting.
Litespark-005 is important because it could support Welireg’s approval beyond the niche indication of Von Hippel-Lindau disease-associated renal cancer to which it’s currently limited. And, with numerous Litespark trials ongoing, Merck & Co needs major wins to justify the $1bn it paid to acquire Welireg’s maker, Peloton Therapeutics.
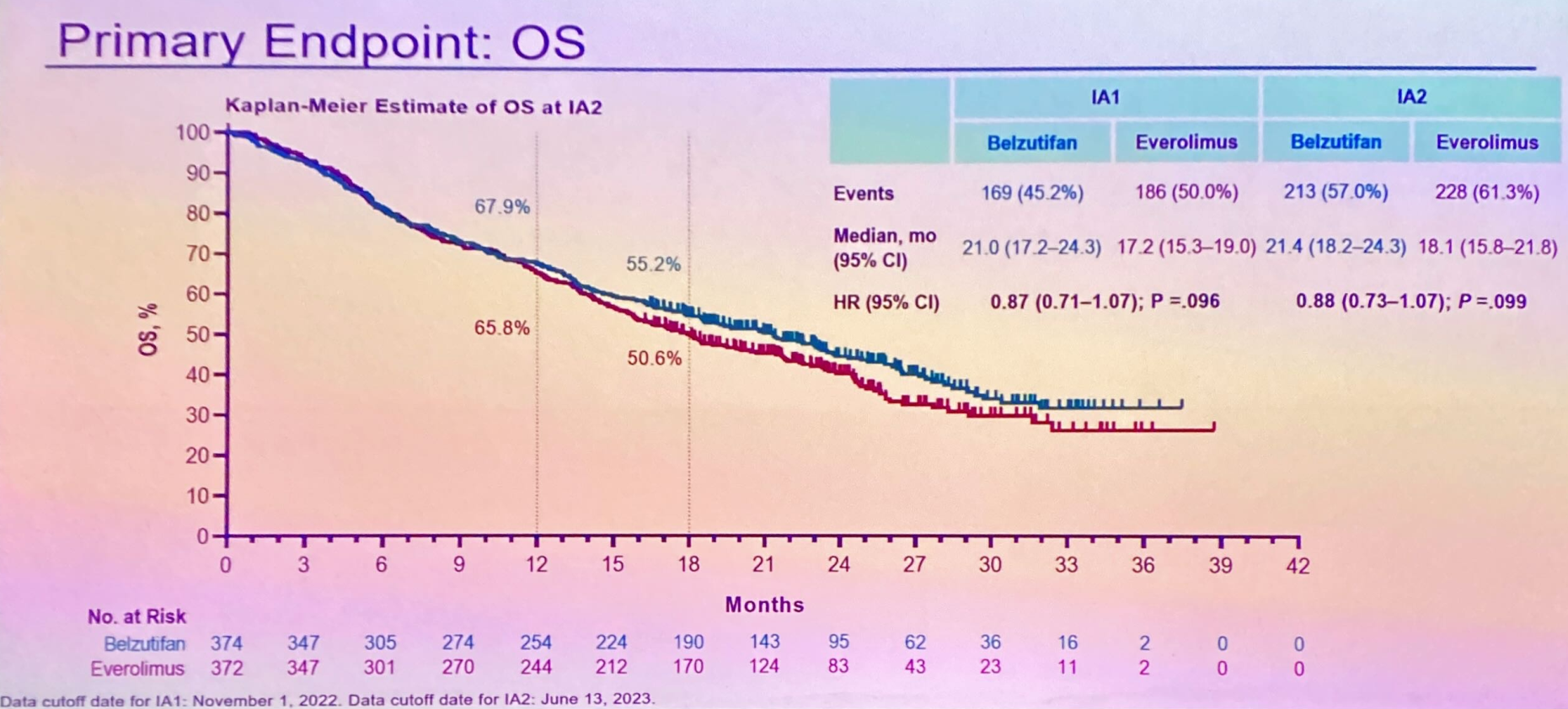
Merck toplined the positive hit on progression-free survival back in August, and ESMO today saw full data for the first time. Litespark-005 tests Welireg, the first approved HIF-2α inhibitor, in second to fourth-line kidney cancer, comparing it against everolimus.
The PFS curves showed a 25% reduction in risk of progression of death, meeting statistical significance at p <0.001 at first interim analysis, a benefit that remained constant at the second blinded review. A significant difference in response rates was also seen, an effect that deepened at second analysis to hit ORRs of 23% for Welireg and 4% for everolimus.
So far so good, but hopes of finding positive signals in overall survival – a key co-primary endpoint known to have been missed at interim – fell on stony ground. The medians have already been hit, and the best that can be said is that a survival “tail” is developing after about 13 months; there’s a 3.3-month median OS benefit, and the 0.88 hazard ratio yielded p=0.099.
Source: Dr Laurence Albiges & ESMO.
Presenting the data, Gustave Roussy’s Dr Laurence Albiges argued that final OS analysis was pending, and highlighted the fact safety looked broadly similar for Welireg and everolimus.
However, the trial’s discussant, the Royal Marsden’s Dr Lisa Pickering, poured cold water on the enthusiasm, saying she might have expected OS to be positive, and was now uncertain whether the result would change meaningfully. As for arguments that PFS alone made this a positive study, she specified: “Improvements in OS are a key goal, therefore we would prefer OS to be positive.”
Not only did she reject a suggestion that everolimus overperformed in Litespark-005, she opined that the drug might not even have been the highest bar for OS. And her doubts extended to the PFS curves, where she drew attention to a lot of early crossing over, and called for more analysis of potential biomarkers that could be more predictive of a positive effect.
Albiges, in turn, argued that Litespark-005 was an open-label trial, and numerous everolimus subjects were thus motivated to discontinue, leading to a lot of censoring in the early part of the analysis.
Despite the doubts Pickering agreed that Welireg seemed “meaningfully active”, and highlighted ongoing adjuvant and front-line trials. In a separate ESMO late-breaker, Dana-Farber’s Dr Toni Choueiri unveiled the uncontrolled Litespark-003 trial, where Welireg plus Exelixis’s Cabometyx yielded ORRs of 70% in treatment-naive and 31% in post-immuno/targeted therapy renal cancer patients.
After all the caveats, Pickering called Litespark-005 a practice-changing study, and said she hoped Welireg would soon be available for these renal cancer patients. That decision will, of course, be down to the regulatory agencies.
Link to OncologyPipeline project
2119













